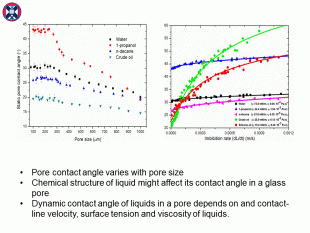
The results indicate that the pore contact angle of a confined liquid in a glass capillary differs from the contact angle measured on a flat glass surface in an open space. Surface tension is not the only dominant factor on contact angle. The static contact angle in a glass pore also varies with glass pore size and liquid chemical structure. Viscosity and surface tension can significantly affect the dynamic pore contact angle. A new empirical correlation was developed based on our experimental data to describe the dynamic pore wetting.
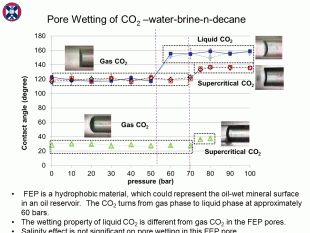
The CO2 phase significantly affects the CO2-fluid contact angle in an oil-wet pore. Supercritical CO2-fluid contact angles are larger than gas CO2-fluid contact angles, but are smaller than liquid CO2-fluid contact angles. However, this significant CO2 phase effect cannot be observed in a water-wet pore.