Post date:
George Serghiou in the School of Engineering together with colleagues from the School of Geosciences and three other leading research institutions in Germany and France have identified a surprising new experimental route to bulk solid solution formation.
Background
A key area of our research is in the synthesis and design of advanced new materials. These new materials are created for specific intended properties which would allow them to perform better than existing materials used in a vast range of industries and settings, from renewable energy to medical technologies.
Researchers working on the synthesis of materials often plan their experiments according to scientific principles which date back nearly 100 years. These governing principles have in particular dictated our understanding on what starting materials can dissolve into each other to form solid solutions.
However, these long-established experimental principles and parameters can be a hindrance when it comes to creating new solid solutions – a pivotal area of research activity from both chemical and technological standpoints.
Ge-Sn materials system
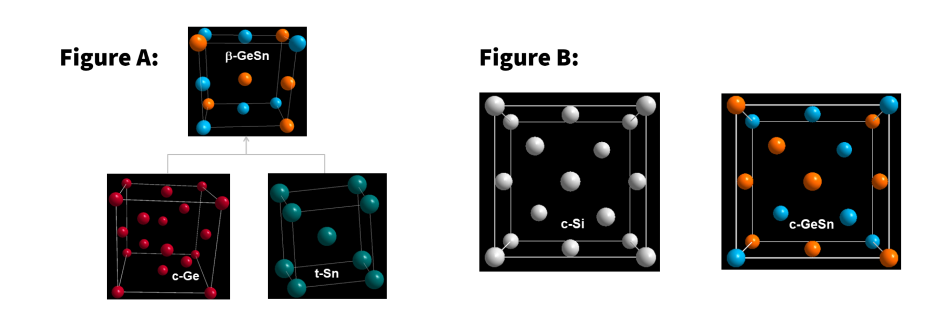
One focus of the Edinburgh researchers is on the Group IVA materials system – a group within the periodic table containing the elements germanium (Ge), tin (Sn), carbon (C), silicon (Si), lead (Pb), and flerovium (Fl).
The Ge-Sn system, in particular, holds promise for advanced new materials particularly useful for optoelectronic applications including laser and optical measurement technologies, photovoltaics and optical fiber communications.
This promise however, can be fulfilled only if researchers are able to dissolve Ge and Sn in the same structure, to create new bulk solid materials. This has previously been difficult because of the low reactivity of these elements with each other.
Up to this point, therefore, researchers have only been able to produce useful materials incorporating the Ge and Sn elements in tiny amounts – as thin films in typically submicron product sizes.
The new route that Dr Serghiou and his colleagues have discovered is surprising because the starting Ge and Sn ingredients dissolve in each other even though they have markedly different structures, electronic characteristics and atomic radii.
Optoelectronic potential
Dr Serghiou explained, “The resulting solid solution created is not only structurally novel but also electronically appealing because it is built from semiconducting Ge and metallic Sn, courtesy of the solid solutions unconventional origins.
“Moreover this solid solution transforms at ambient conditions (in other words, at room temperature and pressure) into the same structure as cubic silicon (Si) – the material driver of the electronics industry, which is found in laptops, smartphones, digital cameras and most other electronic devices. Only now all the Si has been replaced with Ge and Sn.
“This is a good thing because – as effective as cubic silicon is for electronics – it is poor for optoelectronics. This is exactly where the complete replacement of Si with Ge and Sn comes in handy, because the latter two in this structure make this bulk material an optoelectronically important one, limited previously to thin film form.”
These findings open up innovative avenues to advanced materials development.
Recognition
Dr Serghiou and colleagues' work in advanced materials production has recently been selected as "one of the most exciting subjects investigated at the European Synchrotron Radiation Facility (ESRF) over the last year”, and will feature in the ESRF's 2021 Highlights Issue.